
What is Abilify?
Abilify is a known to be killer drug that can have a dangerous withdrawal and that due to its dependency and known possible organ damage never should have been brought to market.
Abilify (Aripiprazole) is a purchased Japanese Compound that was brought to market in 2002 by Bristol Myers Squibb. It is used to treat those diagnosed with mental illnesses. It is important to note that diagnoses can be incorrect.
Abilify can have a very dangerous withdrawal and it can damage many body parts including: lungs, kidneys, reproductive organs, liver, eyes, and adrenal glands. It can also cause diabetes. Many of the problems are exacerbated once the drug is stopped. Unmitigated adrenaline runs can happen which can cause erratic and dangerous behavior and severe insomnia. This information is known by the manufacturer and the FDA but it is suppressed and not provided to patients, providers or family in a cohesive manner.
Detailed pharmacology can be found in the FDA studies. It is important to note that “in general the mechanism of action of antipsychotic medications is unknown,” according to the FDA.
Abilify is a multi-billion dollar franchise drug that sold for $30 per pill a day and grew to be one of the highest selling drugs by revenue in the US in 2014. It found favor because it did not have the sedation like other medications and was marketed for not producing weight gain. Both of these are misleading and the sales of the drug benefitted from this.
The efficacy of many mental health drugs and the need for daily medicating so many people has legitimately been brought into questions. Abilify does not sedate but rather has an anesthesia component to it and although it may not cause immediate weight gain like many mental health drugs, it causes problematic weight gain over time. Abilify is known to cause damage to the body in many areas and can have a very dangerous withdrawal. There can be a host of problems after the drug is ceased because it damages the body and the damage can become more acute once the drug is stopped and because it leaves a poison in the body.
Abilify (Aripiprazole is the active ingredient in Abilify) was initially targeted at individuals suffering from schizophrenia and then individuals categorized as bipolar, it was then illegally pushed on the elderly, marketed as a boost for depression medication, touted as a relief drug to those suffering from cocaine withdrawal, and now marketed to children who suffer from a variety of maladies. Each product line or patient group addition extended the patent life of the drug and kept the price artificially high.
Bristol Myers Squibb has been fined over $500 million for its marketing practices against Abilify and the drug now has a black box warning, yet it is still being prescribed and there is no real cohesive effort to warn the public, train providers, or help survivors.
Methodology for Abilify Survivors’ Guide
The Abilify Survivors Guide used primary research from a survivor who suffered withdrawal multiple times as well as secondary research from noteworthy and well respected professional journals. In additional, interviews and information was collected from multiple physicians. Statistical modeling and techniques were also employed.
Why is an Abilify Survivors Guide Necessary?
Research has shown that Abilify withdrawal can lead to suicidal ideations and even death and the drug itself can cause death and bodily damage. This information is not widely known as previous warnings have been ceased and as the behavior is likely to be construed as mental illness when it very well could be a ramification from the drug. Up until 2012, The Bristol Myer Squibb website stated Detox when ceasing Abilify. Many providers did not take this warning seriously and the manufacturer did not make provisions with detox centers for this.
The primary research survivor suffered withdrawal from Abilify four times, once with immediate cessation, twice with tapering off, and one time a hybrid.
The tapering off was the only method followed through and it was physically and psychologically painful and the danger of self-harm was present.
The survivor was diagnosed by a board certified neurologist and a board certified psychiatrist who is also a neurologist as having suffered Abilify withdrawal. In addition the psychiatrist diagnosed aripiprazole poisoning and lack of access to medical care. The aripiprazole poisoning is key because the symptoms present during the withdrawal can reappear and there can be organ damage.
The survivor suffered the withdrawal even while medicated on other psychiatric medications, although it was not as drastic.
During the immediate cessation withdrawal the survivor had severe abdominal pain, sensitivity to light and smells and migraine type headaches and at times could not stop crying.
In addition, after being on the medication for over five years, the survivor developed severe insomnia and had to be medicated to sleep. The survivor was also prescribed a non-therapeutic dosage of a neuroleptic for sleep once she began Abilify, a possible indication that it was known from the beginning that Abilify impairs sleep.
During the tapering off withdrawal, the survivor suffered adrenal rushes, insomnia, severe pain, acute senses, and weakness, and a board certified psychiatrist stated that this could induce suicidal contemplation or action.
The FDA Animal Studies/Trials for Abilify prove the drug has a withdrawal and is harmful and that the degree of harm was never fully explored or reported. The duty to protect patients from harm was abdicated. The duty to provide providers complete information was neglected. Abilify in many ways was positioned as a wonderful new breakthrough that did not sedate and did not cause weight gain- and the general public was left with the impression that the drug does not harm.
Most practioners and emergency rooms are unaware of the dangerous withdrawal, pain, and body damage caused by Abilify and due to the lack of information, long half-life of the drug (it can take 20-25 days before the last dose leaves the body due to the 72-96 hour half-life) and as a result, if the survivor has a history of mental illness will likely just attribute the behavior to mental illness instead of a very real ramification from the drug Abilify.
There used to be warnings for withdrawal in that the Bristol Myers Squibb website up until early 2012 stated that 30 days in detox was the noted treatment during cessation of the drug Abilify. The survivor studied suffered withdrawal during 2011 and was informed of this, yet no provisions were made by the manufacturer as the survivor searched for detox places equipped to handle Abilify withdrawal and could not find any. Bristol Myers Squibb was contacted in early 2012 for a list of detox centers and the request went unanswered.
The survivor’s reaction to Abilify cessation may not be typical or common but it is a known event. Utilizing the survivor’s characteristics, middle aged female, red hair (red heads can have unique sensitivities to anesthesia as evidenced by work done by Dr. Gary Loyd and as the original pharmaceutical studies acknowledge, Abilify is an analgesic), on the drug for 5 plus years, a modeling of the potential number of survivors per year is as follows:
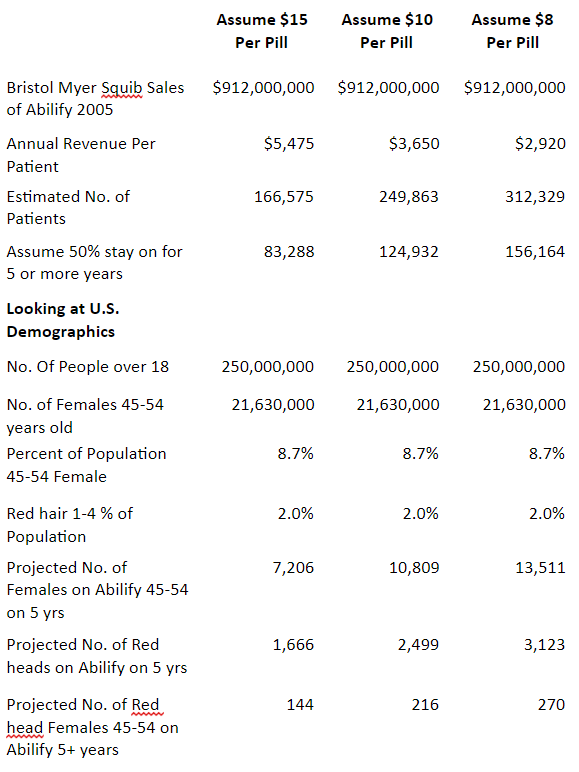
Why is the Abilify Survivors’ Guide Available Now?
The Mayo Clinic research department has been provided with the research and some of the investment community has also been provided with the research and there are now also practitioners that are willing to help. Former CEO John Noseworthy of the Mayo Clinic and Activist Carl Icahn have been made aware of the issues with Abilify. Most Board members and key members of Bristol Myers Squibb upper management have been made aware of the issues the survivor suffered with Abilify.
There are success stories of people getting off Abilify safely. Although the survivor studied in this research may be an extreme case, it is important that is made known that this can happen and steps be taken to provide for care. Bristol Myers Squibb initially provided a public warning in that it stated 30 days in detox when ceasing Abilify, but after being contacted by the survivor after the survivor suffered withdrawal , Bristol Myers Squibb actively took steps to remove the instructions of 30 days in detox from its website in 2012. It has however, introduced a form of Abilify that monitors whether it is ingested. This suggests that ceasing medication may be a hazard.
In addition, while much of the political landscape focuses on the cost of medications, this is not the core issue. The efficacy and safety of medications like Abilify are very real issues and issues that the FDA let slip by. Unlike Canada, and much of the EU, the US FDA is not mandated toward safety and as a result medications that are a violation of the Hippocratic Oath are widely prescribed for daily usage. This guide hopes to point that out and promote greater safety. When discontinuing a drug is dangerous, forcing its continuation or monitoring its continuation is wrong.